immune system
Gut Health and Immunity: The 70% Connection
Discover why 70-80% of your immune system lives in your gut. Learn about GALT, how your microbiome trains immunity, and evidence-based strategies to strengthen the gut-immune connection.
Here is a statistic that surprises most people: somewhere between 70% and 80% of your entire immune system resides in and around your gut. That means the health of your digestive tract directly determines how well your body fights off infections, manages inflammation, and even prevents autoimmune conditions.
For years, we thought of the gut as simply a food-processing tube. But modern research in immunology and microbiology has revealed something far more profound — your gut is essentially immune system headquarters. The trillions of bacteria living in your intestines are not passive residents. They actively train, regulate, and support your immune cells every single day.
This is not just academic theory. Studies published in Nature, Cell, and the Journal of Clinical Investigation consistently show that disruptions in gut health — from poor diet, chronic stress, or antibiotic overuse — lead to measurable weakening of immune function. And the reverse is equally true: improving your gut health can meaningfully strengthen your immunity.
If you want to understand how your gut and immune system work together, you are in the right place. We will walk through the science of GALT, explain how gut bacteria train your immune cells, cover what goes wrong during dysbiosis, and give you a clear action plan backed by current research. For a broader look at gut health, see our complete gut health guide. And if you are focused on overall immune support, our guide to boosting your immune system naturally is a great companion read.
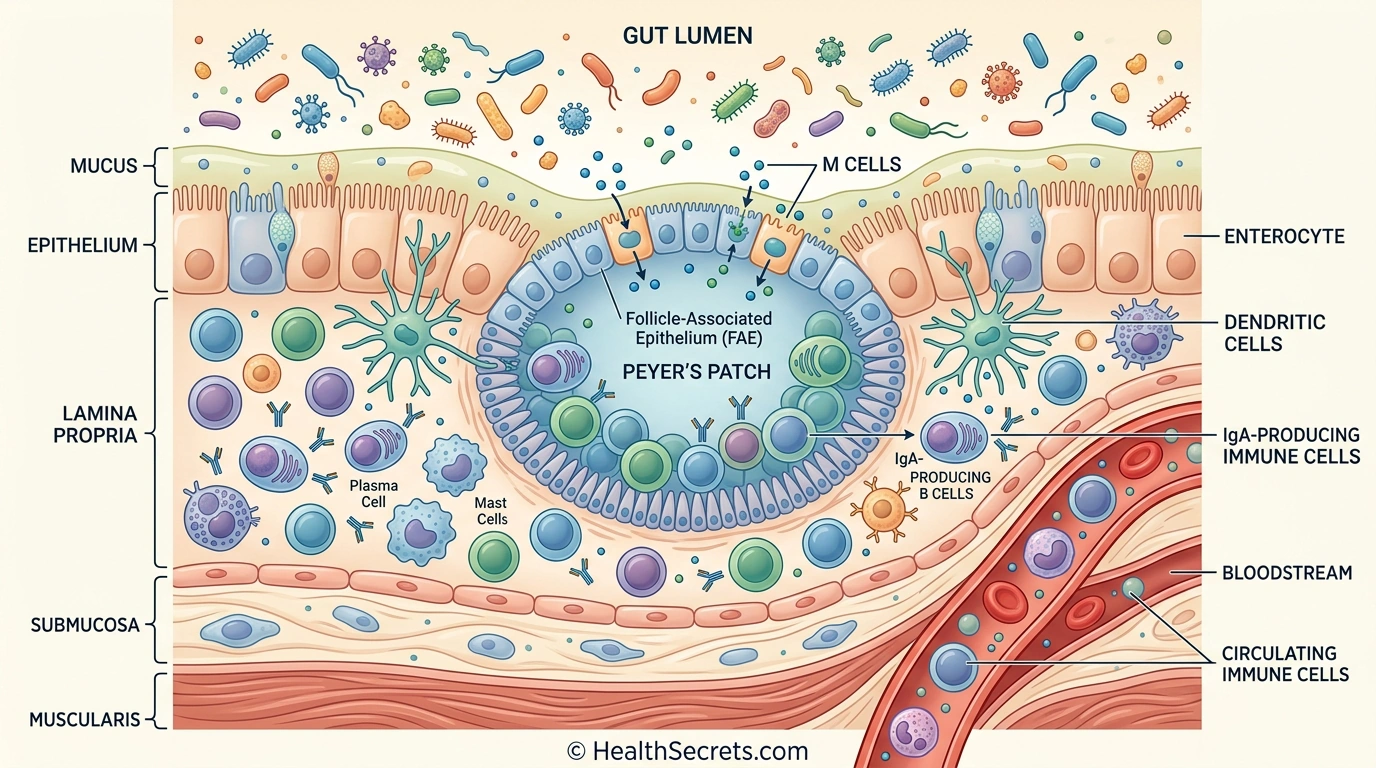
- About 70–80% of your immune cells reside in gut-associated lymphoid tissue (GALT), making the gut your body's largest immune organ.
- GALT includes Peyer's patches, M cells, dendritic cells, and IgA-producing B cells that constantly monitor and respond to threats entering through your digestive tract.
- Your gut microbiome actively trains immune cells to distinguish harmful pathogens from harmless food particles and beneficial bacteria.
- Short-chain fatty acids (SCFAs) produced by beneficial gut bacteria — especially butyrate — fuel intestinal cells, reduce inflammation, and support regulatory T cell development.
- Dysbiosis (microbial imbalance) weakens gut barrier integrity, increases intestinal permeability, and triggers chronic low-grade inflammation linked to autoimmune conditions and frequent infections.
- A diet rich in fermented foods, prebiotic fiber, and polyphenols supports both microbiome diversity and immune resilience.
- Targeted probiotics containing strains like Lactobacillus rhamnosus GG and Bifidobacterium lactis have clinical evidence supporting reduced respiratory infection rates.
- Gut healing follows the 5R protocol: Remove triggers, Replace digestive support, Reinoculate with probiotics, Repair the gut lining, and Rebalance with lifestyle changes.
What Is the Gut-Immune Connection and Why Does 70% of Your Immune System Live in Your Gut?
The gut-immune connection refers to the intimate, bidirectional relationship between your gastrointestinal tract and your immune system. Approximately 70–80% of your immune cells reside in gut-associated lymphoid tissue (GALT), a specialized network of immune structures embedded throughout your intestinal wall. This makes your gut the single largest immune organ in your body — and the frontline of your defense against disease.
The reason so much immune tissue concentrates in the gut is straightforward: your intestines represent the body's largest interface with the outside world. With a surface area of roughly 300–400 square meters, your gut encounters an enormous volume of foreign material daily — food particles, bacteria, viruses, and environmental toxins. The immune system needs to be right there, constantly sampling, monitoring, and deciding what to tolerate and what to attack.
GALT comprises several key structures that work together as a coordinated defense network:
| GALT Component | Location | Primary Function | Why It Matters for Immunity |
|---|---|---|---|
| Peyer's Patches | Small intestine wall | Sample antigens from gut lumen and initiate immune responses | Train T and B cells; produce IgA antibodies |
| M Cells (Microfold Cells) | Epithelial layer over Peyer's patches | Transport antigens from gut lumen to immune cells | Enable immune surveillance of gut contents |
| Dendritic Cells | Lamina propria and Peyer's patches | Capture antigens and present them to T cells | Bridge innate and adaptive immunity; determine tolerance vs. attack |
| Intraepithelial Lymphocytes | Between intestinal epithelial cells | Rapid response to barrier breaches | First responders when pathogens penetrate the gut lining |
| IgA-Producing B Cells | Lamina propria | Secrete IgA antibodies into gut lumen | Neutralize pathogens without triggering inflammation |
IgA is especially remarkable — it is the most abundant antibody your body produces, and it works by neutralizing pathogens and toxins in a non-inflammatory way, essentially keeping the peace while maintaining protection. Research published in Trends in Immunology (2024) confirms that GALT B cells are chronically activated by the intestinal microbiota throughout life, continuously adapting your immune defenses.
How Does Your Gut Microbiome Train and Regulate Immune Function?
Your gut microbiome — the trillions of bacteria, fungi, and other microorganisms living in your intestines — acts as your immune system's primary training partner. These microbes educate immune cells to distinguish between harmful pathogens and harmless substances, produce anti-inflammatory metabolites like short-chain fatty acids, and maintain the gut barrier that prevents dangerous molecules from entering your bloodstream.
The training process starts at birth. Infants born vaginally receive their first bacterial colonization from the birth canal, and breastfeeding provides both beneficial bacteria and prebiotics that feed them. The first three years of life represent a critical window where microbial diversity shapes immune development — children with more diverse microbiomes tend to develop stronger, better-calibrated immune systems.
Throughout adulthood, beneficial bacteria continue to educate your immunity through several key mechanisms:
How Do Gut Bacteria Produce Short-Chain Fatty Acids That Fuel Immunity?
When beneficial bacteria ferment dietary fiber, they produce short-chain fatty acids (SCFAs) — primarily butyrate, acetate, and propionate. Butyrate is especially important: it serves as the primary fuel source for colonocytes (intestinal lining cells), strengthens tight junctions between cells, and promotes the development of regulatory T cells (Tregs) that prevent excessive immune reactions. A 2023 study in Nature Reviews Immunology demonstrated that SCFA-producing bacteria directly modulate both local gut immunity and systemic inflammatory responses throughout the body.
How Does the Gut Barrier Serve as Your First Line of Immune Defense?
Your gut barrier is a multi-layered defense system consisting of the mucus layer, the epithelial cell layer sealed by tight junction proteins, the immune cell-rich lamina propria, and the microbiome itself. Together, these layers allow nutrients to pass through while blocking pathogens, toxins, and undigested food particles. When tight junctions become compromised — a condition often called "increased intestinal permeability" — bacteria and their byproducts can leak into the bloodstream, triggering chronic immune activation and systemic inflammation.
How Do Gut Bacteria Influence Immunity Beyond the Digestive Tract?
The gut-immune axis is bidirectional and systemic. Bacterial metabolites enter the bloodstream and influence immune cells in distant organs — the lungs (gut-lung axis), the brain (gut-brain axis), and the skin. Research has shown that gut bacteria influence NK cell activity, macrophage function, and T cell differentiation throughout the body. This explains why gut dysbiosis is linked to conditions as varied as asthma, depression, and eczema — all involving immune dysregulation far from the digestive tract.
What Are the Key Benefits of a Healthy Gut-Immune Connection?
Maintaining a healthy gut-immune connection provides measurable, clinically documented benefits that extend well beyond digestive comfort. A balanced microbiome supports reduced infection risk, better vaccine responses, lower chronic inflammation, and improved immune tolerance — which means fewer allergies and reduced autoimmune risk.
Does a Diverse Microbiome Reduce Your Risk of Infections?
Yes — multiple meta-analyses confirm that people with diverse gut microbiomes and those taking targeted probiotics experience 20–30% fewer respiratory infections. Beneficial bacteria compete with pathogens for resources (competitive exclusion), produce antimicrobial compounds, and enhance the function of NK cells and macrophages. Regular consumption of fermented foods contributes to this microbial diversity.
Can Gut Health Prevent Allergies and Autoimmune Conditions?
A well-trained immune system knows when to mount an attack and when to stand down. Gut bacteria are essential for developing immune tolerance — the ability to recognize harmless substances like food proteins and pollen without overreacting. Dysbiosis disrupts this tolerance, which is why researchers have linked impaired gut health to conditions including allergies, asthma, rheumatoid arthritis, and inflammatory bowel disease.
How Do Probiotics Improve Vaccine Effectiveness?
Several clinical trials demonstrate that specific probiotic strains — particularly Bifidobacterium lactis and Lactobacillus casei — can enhance antibody production following vaccination. This is especially relevant for older adults, whose immune responses to vaccines naturally decline with age. Supporting gut health before and after vaccination may help your body mount a stronger, longer-lasting protective response.
Does Gut Barrier Integrity Protect Against Chronic Inflammation?
Absolutely. When the gut barrier is intact, the immune system remains in a calm, surveillance state. When it is compromised, bacterial endotoxins (particularly lipopolysaccharide, or LPS) enter the bloodstream and trigger chronic low-grade inflammation — a condition linked to heart disease, type 2 diabetes, obesity, and cognitive decline. Supporting gut barrier health is one of the most effective ways to reduce systemic inflammation.
What Happens When Gut Dysbiosis Weakens Your Immune System?
Dysbiosis — an imbalance in your gut microbiome characterized by reduced bacterial diversity, loss of beneficial species, and overgrowth of harmful bacteria — directly undermines immune function. It weakens the gut barrier, reduces protective SCFA production, triggers chronic inflammation, and impairs immune cell training, leaving you more vulnerable to infections, allergies, and autoimmune conditions.
Common causes of dysbiosis include antibiotic overuse, diets high in processed foods and sugar, chronic stress, inadequate sleep, and environmental toxin exposure. Even medications like proton pump inhibitors (PPIs) and NSAIDs can disrupt the microbiome.
| Dysbiosis Effect | How It Weakens Immunity | Related Conditions |
|---|---|---|
| Reduced SCFA production | Less fuel for intestinal cells; reduced Treg development; weakened barrier | IBD, chronic inflammation, metabolic syndrome |
| Increased intestinal permeability | Bacterial endotoxins enter bloodstream; chronic immune activation | Autoimmune diseases, food sensitivities, systemic inflammation |
| Loss of competitive exclusion | Pathogenic bacteria colonize more easily; reduced antimicrobial compound production | Recurrent infections, C. difficile, SIBO |
| Impaired immune training | Less microbial diversity means poorly calibrated immune responses | Allergies, asthma, eczema, autoimmunity |
| Chronic inflammatory signaling | Elevated pro-inflammatory cytokines (IL-6, TNF-α, CRP) | Heart disease, diabetes, depression, neurodegeneration |
The vicious cycle is particularly concerning: dysbiosis weakens immunity, which further destabilizes the microbiome, which further weakens immunity. Breaking this cycle requires a deliberate, multi-pronged approach — which brings us to practical strategies.
How Can You Strengthen Your Gut-Immune Connection?
Strengthening your gut-immune connection follows the well-established 5R protocol: Remove triggers that damage the gut, Replace digestive support, Reinoculate with beneficial bacteria, Repair the gut lining, and Rebalance through sustained lifestyle changes. Most people notice improvements in digestion within 4–8 weeks, though full immune restoration may take 3–6 months.
- Phase 1 — Remove (Weeks 1–2): Eliminate processed foods, excess sugar, artificial sweeteners, and excess alcohol. Identify and temporarily remove suspected food sensitivities (common triggers include gluten and dairy). Reduce unnecessary NSAID use where possible.
- Phase 2 — Replace (Weeks 1–4): Support digestion with adequate stomach acid and digestive enzymes if needed. Ensure you are eating slowly, chewing thoroughly, and not drinking excessive liquids with meals.
- Phase 3 — Reinoculate (Weeks 2–8): Introduce probiotic-rich fermented foods daily — yogurt, kefir, sauerkraut, kimchi, and miso are excellent choices. Consider a targeted probiotic supplement with clinically studied strains (see product recommendations below). Add prebiotic foods like garlic, onions, leeks, asparagus, and slightly green bananas to feed beneficial bacteria.
- Phase 4 — Repair (Weeks 4–12): Support gut lining repair with L-glutamine (5–10g daily), collagen peptides or bone broth, omega-3 fatty acids, and vitamin D (if deficient). Zinc carnosine (75mg twice daily) has clinical evidence for supporting mucosal healing.
- Phase 5 — Rebalance (Ongoing): Maintain the gains with consistent stress management, 7–9 hours of sleep per night, regular moderate exercise, and an ongoing whole-foods diet rich in fiber and fermented foods. For more on this protocol, see our gut-healing foods guide.
What Foods and Lifestyle Changes Best Support Gut-Immune Health?
The most powerful tool for gut-immune health is a diverse, whole-foods diet rich in fiber, fermented foods, and polyphenols — combined with consistent stress management, adequate sleep, and regular moderate exercise. Research shows that eating 30 or more different plant foods per week significantly increases microbiome diversity.
- Probiotic foods deliver beneficial bacteria directly: yogurt with live cultures, kefir, raw sauerkraut, kimchi, kombucha, miso, and tempeh. Aim for at least one serving of fermented food daily.
- Prebiotic foods feed your existing beneficial bacteria: garlic, onions, leeks, asparagus, artichokes, bananas (especially slightly green), oats, apples, berries, flaxseeds, and chia seeds. Target 25–35g of fiber daily from diverse sources.
- Polyphenol-rich foods support bacterial diversity: berries, pomegranate, green tea, dark chocolate (70%+ cacao), olive oil, and colorful vegetables.
Gut-healing foods support barrier repair: bone broth (rich in glutamine and collagen), omega-3-rich fatty fish (2–3 servings weekly), ginger, and turmeric.
Foods to minimize: Processed foods with emulsifiers and additives, excess sugar, artificial sweeteners, trans fats, excess alcohol, and highly processed seed oils — all of which research links to gut barrier damage and microbial imbalance.
Lifestyle factors are equally important. Chronic stress alters gut bacteria composition and increases intestinal permeability — daily stress management through meditation, yoga, or deep breathing is essential. Poor sleep disrupts the microbiome and impairs immune function; aim for 7–9 hours on a consistent schedule. Moderate exercise (30 minutes most days) increases bacterial diversity, but excessive intense exercise can temporarily suppress immunity. After antibiotic courses, prioritize rebuilding your microbiome with probiotics and fiber-rich foods.
What Should You Do First to Improve Your Gut-Immune Health?
Start with the highest-impact, lowest-effort changes: add one serving of fermented food daily, increase your fiber intake to 25–35g from diverse plant sources, and remove the most damaging processed foods. These three changes alone can measurably shift your microbiome composition within two to four weeks, based on published research.
Week 1–2 (Foundation):
- [ ] Add one serving of fermented food daily (yogurt, kefir, sauerkraut, or kimchi)
- [ ] Increase fiber from diverse plant foods (aim for 25g daily minimum)
- [ ] Remove or reduce processed foods, excess sugar, and artificial sweeteners
- [ ] Begin a daily stress management practice (even 5 minutes of deep breathing)
Week 3–4 (Build):
- [ ] Incorporate specific prebiotic foods — garlic, onions, asparagus, bananas
- [ ] Start a quality probiotic supplement if needed (see recommendations below)
- [ ] Prioritize 7–9 hours of consistent sleep
- [ ] Add 30 minutes of moderate exercise on most days
Week 5–8 (Repair):
- [ ] Add gut-repair nutrients if you suspect leaky gut — L-glutamine, collagen, omega-3s
- [ ] Try eating 30+ different plant foods per week for maximum diversity
- [ ] Monitor your digestion, energy, and illness frequency for improvements
Month 3–6 (Sustain):
- [ ] Maintain dietary changes as permanent habits
- [ ] Reassess supplement needs
- [ ] Consider functional testing (comprehensive stool test) if chronic issues persist
- [ ] Consult a healthcare provider for persistent symptoms
References
- Wiertsema SP, van Bergenhenegouwen J, Garssen J, Knippels LMJ. The Interplay between the Gut Microbiome and the Immune System in the Context of Infectious Diseases throughout Life and the Role of Nutrition in Optimizing Treatment Strategies. Nutrients. 2021;13(3):886. Source
- Zheng D, Liwinski T, Elinav E. Interaction between microbiota and immunity in health and disease. Cell Research. 2020;30(6):492-506. Source
- Spencer J, Bemark M. Gut-associated lymphoid tissue: a microbiota-driven hub of B cell immunity. Trends in Immunology. 2024;45(3):211-223. Source
- Agace WW, McCoy KD. Regionalized Development and Maintenance of the Intestinal Adaptive Immune Landscape. Immunity. 2017;46(4):532-548. Source
- Spencer J, Sollid LM. The human intestinal B-cell response. Mucosal Immunology. 2016;9(5):1113-1124. Source
- Yoo JY, Groer M, Dutra SVO, Sarkar A, McSkimming DI. Gut Microbiota and Immune System Interactions. Microorganisms. 2020;8(10):1587. Source
- Fung TC, Olson CA, Hsiao EY. Interactions between the microbiota, immune and nervous systems in health and disease. Nature Neuroscience. 2017;20(2):145-155. Source
- Belkaid Y, Hand TW. Role of the microbiota in immunity and inflammation. Cell. 2014;157(1):121-141. Source
- Hao Q, Dong BR, Wu T. Probiotics for preventing acute upper respiratory tract infections. Cochrane Database of Systematic Reviews. 2015;(2):CD006895. Source
- Wastyk HC, Fragiadakis GK, Perelman D, et al. Gut-microbiota-targeted diets modulate human immune status. Cell. 2021;184(16):4137-4153.e14. Source
- Bischoff SC, Barbara G, Buurman W, et al. Intestinal permeability — a new target for disease prevention and therapy. BMC Gastroenterology. 2014;14:189. Source
- Kamada N, Seo SU, Chen GY, Núñez G. Role of the gut microbiota in immunity and inflammatory disease. Nature Reviews Immunology. 2013;13(5):321-335. Source
Frequently asked questions
Is it true that 70% of your immune system is in your gut?
Yes. Research confirms that approximately 70–80% of the body's immune cells reside in gut-associated lymphoid tissue (GALT). This includes Peyer's patches, IgA-producing B cells, T cells, dendritic cells, and other immune structures embedded in the intestinal wall. The gut is your body's largest immune organ because it encounters more foreign antigens daily than any other tissue.
What is GALT and why is it important for immunity?
GALT stands for gut-associated lymphoid tissue — a network of immune structures throughout your intestinal wall. It includes Peyer's patches, isolated lymphoid follicles, M cells, and the lamina propria. GALT constantly samples gut contents, trains immune cells to distinguish threats from harmless substances, and produces IgA antibodies that neutralize pathogens.
Can probiotics actually strengthen your immune system?
Yes, specific probiotic strains have clinical evidence for immune support. Lactobacillus rhamnosus GG reduces respiratory infections by 20–30% in children, and Bifidobacterium lactis enhances NK cell activity and vaccine responses in older adults. The key is using strains with actual clinical evidence at adequate doses (10–50 billion CFU daily).
What is leaky gut and how does it affect immunity?
Leaky gut (increased intestinal permeability) occurs when tight junction proteins between intestinal cells become compromised, allowing bacteria, toxins, and undigested food particles to enter the bloodstream. This triggers chronic immune activation and systemic inflammation, which is linked to autoimmune conditions, food sensitivities, and increased infection susceptibility.
How long does it take to improve gut-immune health?
Most people notice digestive improvements within 2–4 weeks of dietary changes and probiotic supplementation. Measurable shifts in microbiome composition occur within 2–6 weeks. However, full gut barrier repair and immune restoration typically takes 3–6 months of consistent effort, and ongoing maintenance is essential for long-term benefits.
What causes gut dysbiosis?
The most common causes of dysbiosis are antibiotic overuse, diets high in processed foods and sugar, chronic psychological stress, inadequate sleep, and certain medications like PPIs and NSAIDs. Environmental factors including pesticide exposure and lack of microbial diversity in modern living environments also contribute to microbial imbalance.
Which probiotic strains are best for immune support?
The most evidence-backed strains for immunity include Lactobacillus rhamnosus GG (reduces respiratory infections), Lactobacillus casei Shirota (enhances NK cell activity), Bifidobacterium lactis BB-12 (improves immune markers and vaccine response), and Saccharomyces boulardii (supports gut immunity during antibiotic use).
What are short-chain fatty acids and why do they matter for immunity?
Short-chain fatty acids (SCFAs) — butyrate, acetate, and propionate — are produced when beneficial gut bacteria ferment dietary fiber. Butyrate fuels intestinal cells, strengthens the gut barrier, reduces inflammation, and promotes regulatory T cell development. Low SCFA production due to dysbiosis is directly linked to weakened immune function and chronic inflammation.
Can improving gut health help with autoimmune conditions?
Emerging research strongly links gut health to autoimmune disease. Increased intestinal permeability may allow molecular mimicry and immune dysregulation that triggers autoimmunity. While healing the gut is not a cure for autoimmune conditions, many patients experience symptom improvement through gut-healing protocols. Always work with a healthcare provider for autoimmune conditions.
What foods are best for gut-immune health?
The best foods include probiotic-rich fermented foods (yogurt, kefir, sauerkraut, kimchi), prebiotic-rich foods (garlic, onions, leeks, asparagus, bananas), polyphenol-rich foods (berries, green tea, dark chocolate), and gut-healing foods (bone broth, omega-3-rich fish, ginger, turmeric). Aim for 30+ different plant foods weekly.