supplements
Metformin for Longevity | Anti-Aging Drug Guide
Can metformin extend lifespan? Explore the TAME trial, AMPK and mTOR mechanisms, evidence for anti-aging benefits, side effects, dosing, and whether this diabetes drug is right for you.
Here's a strange one for you. A diabetes drug—cheap, generic, been around since the 1950s—might slow down aging itself.
Metformin. You've probably heard the name bouncing around longevity circles, biohacker podcasts, and maybe even your doctor's office. The buzz started when researchers noticed something genuinely bizarre: type 2 diabetics taking metformin were outliving healthy people without diabetes. That's not supposed to happen.
That observation kicked off a wave of research into metformin longevity that's still building today. Animal studies showed lifespan extension. Cellular research revealed metformin activating the same pathways as caloric restriction—one of the most robust anti-aging interventions we know. And now, the TAME trial (Targeting Aging with Metformin) aims to be the first clinical study ever to target aging as a treatable condition.
But hold on. Before you start Googling "how to get metformin without diabetes"—this is a prescription medication. Not a supplement you grab off Amazon. It has real side effects, real contraindications, and the longevity evidence in healthy humans? It doesn't exist yet. Not from completed trials, anyway.
Want the bigger picture on living longer? Start with our comprehensive longevity and anti-aging guide. If you're curious about cellular mechanisms, our guide on telomeres and aging covers the fundamentals. And for the most powerful free longevity tool available, check out exercise for longevity.
- Metformin is a prescription diabetes drug (not a supplement) that activates AMPK and inhibits mTOR—the same longevity pathways triggered by caloric restriction and exercise
- Observational data shows diabetics on metformin may live longer than non-diabetics, but this doesn't prove causation—confounding factors exist
- The TAME trial is the first clinical study designed to target aging itself, testing metformin in 3,000 non-diabetic adults aged 65–79, with results expected in the late 2020s
- No completed randomized controlled trials exist testing metformin for longevity in healthy humans—the evidence gap is significant
- Common side effects include GI distress (20–30% of users), vitamin B12 deficiency with long-term use, and rare but serious lactic acidosis
- Berberine, an over-the-counter plant compound, activates similar AMPK pathways and may serve as a natural alternative for those who can't access or tolerate metformin
- Lifestyle interventions—exercise, fasting, quality sleep—activate the same longevity pathways more powerfully than metformin, with zero side effects
- People with prediabetes, insulin resistance, or metabolic syndrome may have the strongest risk-benefit case for metformin under medical supervision
What Is Metformin and Why Has It Become a Longevity Drug Candidate?
Metformin is a biguanide medication derived from French lilac (Galega officinalis), FDA-approved in 1994 for type 2 diabetes. It's the most prescribed diabetes drug worldwide with over 150 million users. Longevity interest exploded after observational studies suggested diabetics taking metformin had lower all-cause mortality than even non-diabetics—a finding that defied expectations and triggered a wave of anti-aging research.
The drug works primarily by reducing glucose production in the liver and improving insulin sensitivity throughout the body. It's generic, remarkably cheap ($4–20 per month), and has over six decades of human safety data—a combination that makes it uniquely attractive for longevity research ([1]).
What made researchers sit up was a 2014 study by Bannister and colleagues. They found that type 2 diabetics on metformin had 15% lower all-cause mortality compared to matched non-diabetic controls. Diabetics typically die younger. So a diabetes drug making diabetics outlive healthy people? Something beyond glucose control was happening.
Animal data added fuel. Studies in C. elegans worms showed lifespan extension up to 40%. Mouse studies demonstrated 5–10% lifespan extension in some strains, with consistent healthspan improvements—better physical function, delayed disease onset ([3]).
But here's the critical distinction: metformin is not a supplement. It requires a prescription, medical monitoring, and has real contraindications. The off-label longevity use remains experimental.
How Does Metformin Potentially Slow Aging at the Cellular Level?
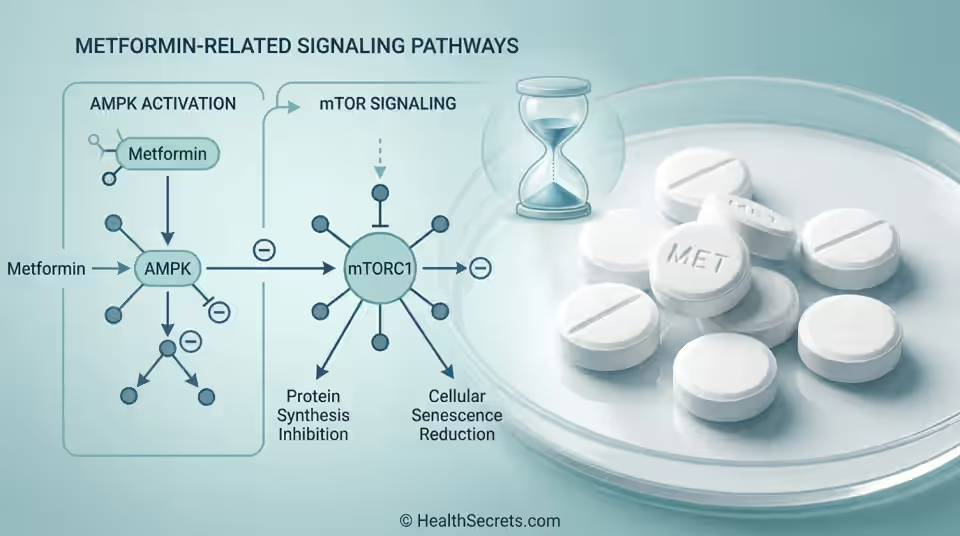
Metformin targets multiple aging hallmarks simultaneously. It activates AMPK (your cellular energy sensor), inhibits mTOR (a growth pathway that accelerates aging when overactive), reduces chronic inflammation, and improves insulin sensitivity. These mechanisms overlap significantly with caloric restriction—one of the most consistently demonstrated lifespan-extending interventions in research ([5]).
How Does AMPK Activation Drive Metformin's Anti-Aging Effects?
AMPK (AMP-activated protein kinase) is your cellular energy sensor. When metformin inhibits complex I of the mitochondrial electron transport chain, the ADP/ATP ratio increases, triggering AMPK activation. This signals "energy is low" even when it isn't—and your cells respond by increasing glucose uptake, enhancing mitochondrial biogenesis, activating autophagy (cellular cleanup), and reducing inflammation ([4]).
AMPK activation essentially mimics the molecular effects of fasting and exercise. That's why metformin is called a caloric restriction mimetic—you get some fasting benefits without actually fasting.
How Does mTOR Inhibition Contribute to Metformin's Longevity Potential?
mTOR (mechanistic target of rapamycin) is a nutrient-sensing pathway that promotes growth and cell division. Overactive mTOR accelerates aging—it's like keeping your foot on the gas constantly. Metformin reduces mTOR activity through both AMPK-dependent and independent mechanisms, leading to increased autophagy, reduced protein synthesis stress, and enhanced cellular resilience ([6]).
Does Metformin Reduce the Chronic Inflammation That Drives Aging?
Chronic low-grade inflammation—sometimes called "inflammaging"—underlies most age-related diseases. Metformin reduces inflammatory markers including IL-6, TNF-alpha, and CRP through AMPK-mediated NF-κB inhibition. Less systemic inflammation translates to slower tissue degradation and reduced disease risk ([26]).
Metformin also modulates the gut microbiome, improving beneficial bacterial populations. Recent research suggests some of metformin's metabolic benefits are actually mediated through these microbiome changes rather than direct cellular effects.
How Well Is Metformin Absorbed and Which Formulation Works Best?
Metformin has moderate oral bioavailability of approximately 50–60%, absorbed primarily in the small intestine. It is not metabolized by the liver and is excreted unchanged by the kidneys—which is why kidney function monitoring is essential. The extended-release (ER/XR) formulation provides slower, steadier absorption with significantly fewer gastrointestinal side effects compared to immediate-release, making it the preferred choice for most longevity-focused protocols.
Immediate-release metformin reaches peak plasma concentration in 2–3 hours and must typically be taken 2–3 times daily with meals. Extended-release hits peak levels in 4–8 hours, allowing once-daily dosing—usually with dinner or the largest meal of the day.
Food significantly affects absorption. Taking metformin with meals both improves absorption and dramatically reduces the GI upset that causes many people to quit. High-fat meals slow absorption but don't reduce the total amount absorbed.
A key pharmacological point: metformin doesn't accumulate in fat tissue. It concentrates primarily in the gut lining, liver, and kidneys. This tissue distribution explains both its mechanism of action (gut microbiome effects, hepatic glucose production) and its primary risk factor (kidney-dependent clearance).
Because kidneys clear 100% of metformin unchanged, anyone with impaired kidney function (eGFR below 30 mL/min) faces dangerous accumulation. This isn't negotiable—kidney function determines whether metformin is safe for you.
How Much Metformin Do Longevity Doctors Typically Prescribe?
Most longevity-focused physicians start at 500mg once daily with dinner, increasing by 500mg every 1–2 weeks as tolerated. Maintenance doses for longevity protocols typically range from 1,000–2,000mg daily. The maximum approved dose is 2,550mg, but most anti-aging protocols stay at 1,500–2,000mg. Extended-release formulations are strongly preferred for tolerability and once-daily convenience.
| Protocol Phase | Daily Dose | Timing | Duration |
|---|---|---|---|
| Starting | 500mg | With dinner | 1–2 weeks |
| Building | 500–1,000mg | With meals | 2–4 weeks |
| Maintenance | 1,000–2,000mg | Once daily (ER) or split | Ongoing |
Monitoring schedule your doctor should follow:
- Baseline: Kidney function (eGFR), liver function, B12 level, fasting glucose, HbA1c
- Every 6 months: Kidney function (eGFR)
- Annually: Vitamin B12 level, liver function, metabolic panel
- As needed: If new symptoms develop or health status changes
Cost reality: Generic metformin runs $4–20 per month without insurance. Extended-release is slightly more but still affordable. Insurance typically covers diabetes/prediabetes indications; off-label longevity use may not be covered, but the out-of-pocket cost is minimal.
One important caveat many longevity enthusiasts miss: temporarily stop metformin 48 hours before and after any procedure using iodinated contrast dye (CT scans with contrast, certain cardiac procedures). The combination increases lactic acidosis risk.
Can You Get Metformin's Anti-Aging Benefits from Natural Alternatives?
You can't get metformin from food—it's a synthetic pharmaceutical. However, several natural compounds and lifestyle interventions activate the same AMPK and mTOR pathways. Berberine, a plant alkaloid found in goldenseal and Oregon grape, is the closest over-the-counter alternative. Exercise and intermittent fasting activate AMPK even more powerfully than metformin, with zero side effects and proven longevity benefits.
Berberine deserves special attention. This plant compound activates AMPK through a mechanism remarkably similar to metformin. Multiple studies show berberine matches metformin for blood glucose reduction in diabetic patients ([14]). Typical dosing: 500mg, 2–3 times daily with meals. Side effects mirror metformin (GI upset), and it's available without prescription for $15–30/month.
But berberine has limitations: less standardized, fewer long-term studies, potential drug interactions (particularly with medications metabolized by CYP enzymes), and no equivalent of the TAME trial planned.
Lifestyle interventions that activate the same pathways:
- Exercise: The most powerful AMPK activator available—free, proven, and with cascading benefits metformin can't match
- Intermittent fasting: Directly activates AMPK and autophagy, inhibits mTOR
- Mediterranean or low-carb diet: Improves insulin sensitivity, reduces inflammation
- Sleep optimization: Poor sleep impairs insulin sensitivity and increases inflammatory markers
- Cold exposure: Activates AMPK and brown adipose tissue
The honest assessment? Master the free stuff first. Exercise, nutrition, sleep, and stress management activate every pathway metformin targets—often more potently. For a detailed protocol, see our exercise for longevity guide.
Is Metformin Safe for Healthy People Considering It for Longevity?
Metformin has an excellent safety record across six decades and over 150 million users—but that data comes almost entirely from diabetic populations. For healthy non-diabetics, the risk-benefit calculation is less clear. Common side effects include GI distress in 20–30% of users, vitamin B12 deficiency with long-term use, and rare but potentially fatal lactic acidosis in people with kidney impairment ([13]).
- Gastrointestinal issues (most common): Nausea, diarrhea, cramping, bloating. Affects roughly one in four people starting metformin. Usually improves within 2–4 weeks. Extended-release formulations and gradual dose titration significantly reduce GI problems.
- Vitamin B12 deficiency (long-term concern): Metformin reduces B12 absorption in the terminal ileum. Long-term use can cause deficiency leading to fatigue, neurological symptoms (numbness, tingling), cognitive issues, and anemia. The UK MHRA now recommends monitoring B12 in all metformin users with risk factors ([11]). Solution: supplement with 500–1,000mcg sublingual B12 and test annually.
- Lactic acidosis (rare but serious): Occurs in approximately 2.4–9 cases per 100,000 patient-years—rare, but potentially fatal. Risk factors: kidney disease, liver disease, heart failure, dehydration, excessive alcohol, acute illness ([12]).
Who should NOT take metformin:
- Kidney disease (eGFR below 30 mL/min)
- Severe liver disease
- Unstable heart failure
- History of lactic acidosis
- Heavy alcohol use
- Pregnancy or breastfeeding
- Upcoming procedures with contrast dye (temporary hold)
What Can Metformin Actually Do for Your Longevity Right Now?
Honestly? We don't know yet—not with certainty. Metformin for longevity in healthy humans is a bet on promising but incomplete evidence. Animal studies show lifespan extension in some models but not others. Observational human data is intriguing but riddled with confounders. The TAME trial will provide the first real answers, but results aren't expected until the late 2020s ([10]).
:::info[What the TAME trial might tell us:] TAME (Targeting Aging with Metformin) is unprecedented—3,000 non-diabetic participants aged 65–79, tracked over 6 years for cardiovascular events, cancer, cognitive decline, and mortality. If metformin delays multiple age-related diseases by even 1–2 years, it validates the entire "target aging itself" paradigm ([1]). :::
But TAME has faced funding challenges. Metformin is generic—no pharmaceutical company profits from proving it works for longevity. Good for accessibility. Bad for research funding.
The emerging uncertainty: A 2026 review in Ageing Research Reviews noted that metformin has "generally not demonstrated its anticipated benefits in most clinical trials in nondiabetic populations," highlighting growing scientific caution about overpromising ([23]).
Realistic expectations if you start metformin today:
- You won't feel different next week or next month
- Benefits (if real) accumulate over years to decades
- You're essentially running a personal experiment with incomplete data
- Lifestyle interventions remain more proven and more powerful
- Individual variation is enormous—genetics, microbiome, and metabolic state all influence response
Who has the strongest case for considering metformin (with medical supervision):
- People with prediabetes or metabolic syndrome (established indication)
- Those with insulin resistance or strong family history of type 2 diabetes
- Aging adults (60+) with metabolic concerns who've already optimized lifestyle
- People who've maximized diet, exercise, and sleep and want additional interventions
Who should probably skip it for now:
- Healthy, metabolically fit adults under 50
- Anyone with contraindications
- Those unwilling to do regular monitoring
- People who haven't optimized lifestyle basics first
The bottom line: metformin is one of the most promising pharmacological longevity candidates we're studying. But it's still experimental for healthy people. Focus on what's proven—move your body, eat well, sleep enough, manage stress. Those interventions activate every pathway metformin targets. And they're free.
What Should You Do First If You're Considering Metformin for Longevity?
Start with lifestyle optimization—exercise, nutrition, sleep, and stress management activate the same longevity pathways metformin targets. If you have metabolic risk factors, schedule a conversation with a doctor knowledgeable about longevity medicine. Get baseline labs (fasting glucose, HbA1c, insulin, kidney function, B12) before any medication discussion. Never self-prescribe metformin.
Phase 1 — Foundation (Weeks 1–4):
- [ ] Optimize exercise routine: 150+ minutes moderate activity weekly, include resistance training
- [ ] Assess metabolic health: fasting glucose, HbA1c, fasting insulin, lipid panel
- [ ] Get kidney function tested (eGFR) and B12 baseline
- [ ] Evaluate diet—Mediterranean or whole-food patterns improve insulin sensitivity
- [ ] Track sleep quality and aim for 7–9 hours consistently
Phase 2 — Evaluation (Weeks 4–8):
- [ ] Review lab results with a healthcare provider
- [ ] Discuss risk-benefit profile based on your metabolic status
- [ ] Research longevity-focused physicians in your area if interested
- [ ] Consider berberine as an OTC alternative if appropriate
- [ ] Read current research on the TAME trial status
Phase 3 — Implementation (If medically appropriate):
- [ ] Start metformin ER at 500mg with dinner (doctor-prescribed only)
- [ ] Increase dose gradually over 4–6 weeks as tolerated
- [ ] Begin B12 supplementation (500–1,000mcg sublingual daily)
- [ ] Schedule 6-month kidney function monitoring
- [ ] Track any side effects and report to your physician
References
- Barzilai, N., et al. 2016. Metformin as a Tool to Target Aging. Cell Metabolism, 23(6), 1060-1065. Source
- Bannister, C.A., et al. 2014. Can people with type 2 diabetes live longer than those without? Diabetes, Obesity and Metabolism, 16(11), 1165-1173. Source
- Martin-Montalvo, A., et al. 2013. Metformin improves healthspan and lifespan in mice. Nature Communications, 4, 2192. Source
- Zhou, G., et al. 2001. Role of AMP-activated protein kinase in mechanism of metformin action. Journal of Clinical Investigation, 108(8), 1167-1174. Source
- Kulkarni, A.S., et al. 2020. Benefits of Metformin in Attenuating the Hallmarks of Aging. Cell Metabolism, 32(1), 15-30. Source
- Kalender, A., et al. 2010. Metformin, independent of AMPK, inhibits mTORC1 in a rag GTPase-dependent manner. Cell Metabolism, 11(5), 390-401. Source
- Zaki, R.A., & El-Osta, A. 2024. Metformin: decelerates biomarkers of aging clocks. Signal Transduction and Targeted Therapy, 9, 319. Source
- Foretz, M., et al. 2014. Metformin: from mechanisms of action to therapies. Cell Metabolism, 20(6), 953-966. Source
- Anisimov, V.N., et al. 2011. Metformin slows down aging and extends life span of female SHR mice. Cell Cycle, 10(24), 4230-4236. Source
- Triggle, C.R., et al. 2022. A Critical Review of the Evidence That Metformin Is a Putative Anti-Aging Drug. Frontiers in Endocrinology, 12, 718942. Source
- MHRA. 2022. Metformin and reduced vitamin B12 levels: new advice for monitoring patients at risk. Drug Safety Update. Source
- Salvatore, T., et al. 2022. Metformin: When Should We Fear Lactic Acidosis? International Journal of Molecular Sciences, 23(15), 8320. Source
Frequently asked questions
Can you buy metformin over the counter for longevity?
No. Metformin is a prescription-only medication in the United States and most countries. You cannot legally purchase it without a doctor's prescription. Online pharmacies selling metformin without prescription are operating illegally and may provide counterfeit or contaminated products. If interested in metformin for longevity, discuss it with a physician who can evaluate your individual risk-benefit profile and prescribe it if appropriate.
Does metformin actually extend human lifespan?
We don't know yet. No completed randomized controlled trial has tested metformin for lifespan extension in healthy humans. Observational data shows diabetics on metformin may have lower mortality than non-diabetics, and animal studies show lifespan extension in some species. But observational data has confounding factors, and animal results don't always translate. The TAME trial aims to answer this definitively, with results expected in the late 2020s.
Is berberine as effective as metformin for anti-aging?
Berberine activates AMPK through a similar mechanism and matches metformin for blood glucose reduction in several studies. However, berberine has less long-term safety data, less standardization, and no equivalent longevity trials planned. It's a reasonable OTC alternative for metabolic health support, but calling it equivalent to metformin for anti-aging overstates the evidence for both compounds.
What is the TAME trial and when will results be available?
TAME (Targeting Aging with Metformin) is the first clinical trial designed to target aging itself as a treatable condition. It will test metformin versus placebo in 3,000 non-diabetic adults aged 65–79 over 6 years, measuring cardiovascular events, cancer, cognitive decline, and mortality. Results are expected in the late 2020s, potentially 2027–2029, though the trial has faced funding delays.
Does metformin interfere with exercise benefits?
This is an active research question. Some studies suggest metformin may blunt certain exercise adaptations—particularly mitochondrial improvements and muscle hypertrophy gains. A 2019 study found metformin attenuated improvements in insulin sensitivity and cardiorespiratory fitness from aerobic exercise training. This doesn't mean you shouldn't exercise on metformin, but it's worth discussing timing and dosing with your doctor.
What are the most common side effects of metformin?
Gastrointestinal issues—nausea, diarrhea, bloating, and abdominal discomfort—affect 20–30% of people starting metformin. These usually improve within 2–4 weeks and are significantly reduced with extended-release formulations and gradual dose increases. Long-term use can cause vitamin B12 deficiency, which requires monitoring and supplementation. Lactic acidosis is extremely rare (2–9 per 100,000 patient-years) but serious.
Should healthy young people take metformin for longevity?
Most longevity experts say no—not yet. For healthy, metabolically fit adults under 50, the evidence doesn't support metformin use. The risk-benefit ratio favors lifestyle interventions (exercise, nutrition, sleep, stress management) which activate the same pathways more powerfully and with no side effects. Metformin may be more reasonable for older adults with metabolic risk factors, under medical supervision.
How long does it take for metformin to show anti-aging benefits?
If metformin has longevity benefits, they accumulate over years to decades—not weeks or months. You won't feel noticeably different in the short term. Metabolic markers (fasting glucose, insulin sensitivity, inflammatory markers) may improve within weeks to months, but actual aging-related outcomes require prolonged use. This is a long-game intervention, not a quick fix.
Can metformin cause kidney damage?
Metformin does not cause kidney damage. However, it is cleared entirely by the kidneys, so existing kidney impairment causes dangerous drug accumulation. People with an eGFR below 30 should not take metformin, and those with eGFR 30–45 require dose adjustment and closer monitoring. Regular kidney function testing (every 6–12 months) is mandatory for all metformin users.
Does metformin reduce cancer risk?
Observational studies consistently show diabetics on metformin have lower rates of several cancers compared to diabetics on other medications. The proposed mechanisms include mTOR inhibition, reduced insulin/IGF-1 signaling, and direct effects on cancer cell metabolism. However, this data comes from diabetic populations and has confounding factors. Whether metformin reduces cancer risk in non-diabetics is unknown and awaits clinical trial data.