longevity
Telomeres and Aging: How to Protect Your DNA
Learn how telomeres and aging are connected. Discover what shortens telomeres (stress, smoking, poor diet) and proven strategies to protect your DNA, including exercise, meditation, and omega-3s.
Have you ever wondered why two people born in the same year can look—and feel—decades apart? The answer may be hiding at the tips of your chromosomes. Telomeres and aging are more deeply connected than most people realize, and the science behind this link earned three researchers a Nobel Prize in 2009.
Telomeres are tiny protective structures at the ends of your chromosomes, often compared to the plastic tips on shoelaces. They prevent your genetic material from fraying and degrading each time a cell divides. But here's the catch: telomeres get a little shorter with every division. Over time, they become critically short, and the cell either stops functioning properly or dies altogether. That process, in a nutshell, is one of the fundamental drivers of biological aging.
The exciting part? Research shows that lifestyle choices—exercise, stress management, diet, and more—can actually influence your telomere length. A landmark study by Dean Ornish and Nobel laureate Elizabeth Blackburn demonstrated that comprehensive lifestyle changes could increase telomerase activity by 30% and even lengthen telomeres over five years.
In this guide, you'll learn exactly what telomeres are, how they connect to aging and disease risk, what shortens them faster, and—most importantly—practical, evidence-based strategies to protect them. Whether you're interested in the complete guide to longevity and anti-aging or curious about how detoxification supports cellular health, understanding telomeres is a foundational piece of the puzzle.
- Telomeres are protective DNA-protein caps on chromosomes that shorten with each cell division, serving as a biological aging clock discovered by Nobel Prize-winning researchers.
- Shorter telomeres are associated with higher risk of cardiovascular disease, type 2 diabetes, certain cancers, and Alzheimer's disease—though correlation doesn't prove causation.
- Chronic stress, smoking, obesity, poor diet, sedentary behavior, and sleep deprivation all accelerate telomere shortening through oxidative stress and inflammation.
- Regular aerobic exercise (30–40 minutes, 5 times per week) is one of the most powerful telomere protectors, with exercisers showing telomere lengths equivalent to 9 years younger.
- The Dean Ornish study showed that comprehensive lifestyle changes—diet, exercise, stress management, and social support—increased telomerase activity by 30% and lengthened telomeres over 5 years.
- Omega-3 fatty acids (1,000–2,000 mg EPA+DHA daily), vitamin D optimization, and a Mediterranean-style diet are all associated with longer telomeres.
- Telomerase, the enzyme that rebuilds telomeres, is a double-edged sword—modest lifestyle-driven increases are safe, but aggressive activation carries cancer risk.
- Commercial telomere testing ($100–$500) exists but has significant limitations including high variability and limited clinical actionability for most people.
What Are Telomeres and Why Do They Matter for Aging?
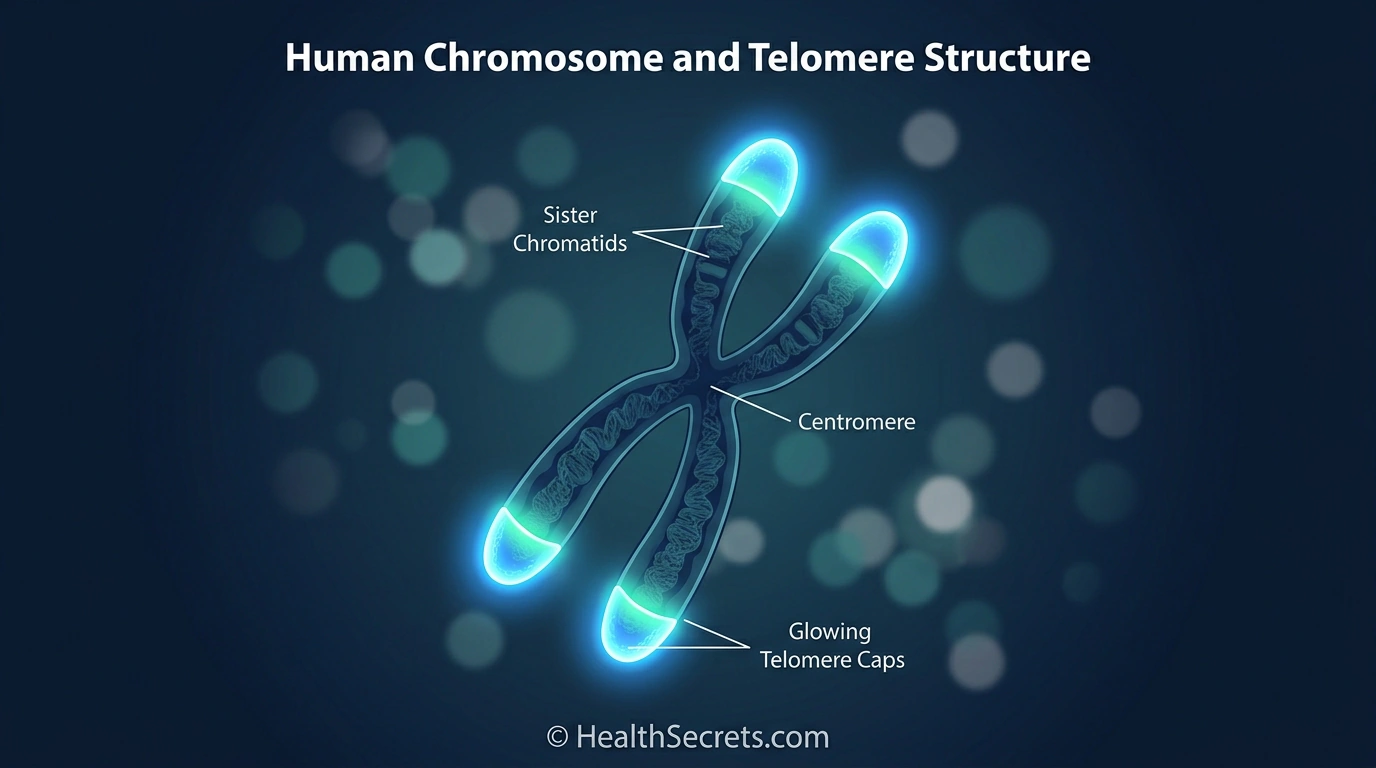
Telomeres are specialized DNA-protein structures located at the ends of each chromosome, and they play a surprisingly important role in how your body ages at the cellular level. Think of them as the plastic aglets on shoelaces—without those tips, the lace unravels. Without telomeres, your chromosomes would fuse together, leading to genomic instability and cell death.
Structurally, human telomeres consist of repetitive DNA sequences—specifically, the six-nucleotide pattern TTAGGG, repeated roughly 1,000 to 2,000 times at each chromosome end. Since you have 46 chromosomes in most cells, that means 92 telomeres per cell, all quietly doing the job of protecting your genetic code.
The story of telomere discovery stretches back decades. In the 1930s, Hermann Muller and Barbara McClintock independently recognized that chromosome ends had special protective properties. Then in the 1970s and 1980s, Elizabeth Blackburn identified the actual DNA sequence of telomeres. In 1984, Blackburn and her graduate student Carol Greider discovered telomerase—the enzyme that rebuilds telomeres. That work, alongside contributions from Jack Szostak, earned all three the [21].
But here's why telomeres matter for aging: each time a cell divides, telomeres lose approximately 50 to 200 base pairs due to what scientists call the "end-replication problem." Most human cells can divide about 40 to 60 times—the so-called Hayflick limit—before telomeres become critically short. At that point, cells enter senescence (a permanent state of growth arrest) or undergo apoptosis (programmed cell death). The result is tissue aging, reduced regenerative capacity, and increased susceptibility to age-related diseases ([1]).
How Does the Telomere-Aging Connection Actually Work?
The connection between telomere shortening and biological aging operates through several interconnected mechanisms—from cellular senescence and disease susceptibility to the complex role of the enzyme telomerase. Understanding these pathways helps explain why telomere length has become one of the most studied biomarkers of aging.
How Do Telomeres Function as a Biological Clock?
Every cell division costs you a small piece of telomeric DNA. Over a lifetime, this steady erosion acts as a kind of molecular countdown timer. Two people who are both 50 years old chronologically can have dramatically different telomere lengths—meaning their biological ages may differ by a decade or more. Shorter telomeres generally correlate with older biological age and greater cellular wear, while longer telomeres suggest younger biological age and better cellular integrity ([11]).
That said, telomeres aren't a perfect crystal ball. Genetics account for roughly 30–80% of your telomere length, and many other factors influence aging beyond this single biomarker. Telomere length is one important piece—not the whole picture.
What Role Does Telomerase Play in Telomere Maintenance?
Telomerase is a remarkable enzyme—a reverse transcriptase that adds TTAGGG repeats back onto telomere ends, essentially counteracting the shortening process. It's highly active in stem cells, germ cells, and certain immune cells, which is why those cells can keep dividing.
But in most adult somatic cells, telomerase is either inactive or barely detectable. And there's a good evolutionary reason for that: 85–95% of human cancers reactivate telomerase to achieve unlimited replication. Telomere shortening actually functions as a tumor suppression mechanism. It's a biological trade-off—shorter telomeres contribute to aging, but suppressing telomerase helps prevent cancer ([14]).
The good news? Modest, lifestyle-driven increases in telomerase activity appear to be safe and beneficial. The Dean Ornish study demonstrated a 30% increase in telomerase activity through diet, exercise, stress management, and social support—without any observed cancer risk increase ([6]).
How Are Shorter Telomeres Linked to Disease?
Research has established associations between shorter telomere length and multiple age-related conditions:
- Cardiovascular disease: Shorter telomeres are associated with heart disease, stroke, and atherosclerosis ([12])
- Type 2 diabetes: Telomere shortening is linked to insulin resistance and metabolic dysfunction ([13])
- Cancer: The relationship is complex—short telomeres increase cancer risk through genomic instability, yet cancer cells reactivate telomerase for survival
- Alzheimer's disease: Shorter telomeres have been observed in patients with neurodegenerative diseases
- All-cause mortality: Meta-analyses show shorter telomere length predicts higher mortality risk
An important caveat: most of this evidence is correlational. Shorter telomeres correlate with disease—they haven't been conclusively proven to cause it.
The relationship likely runs both ways: poor health shortens telomeres, and short telomeres contribute to declining health.
What Are the Key Benefits of Protecting Your Telomeres?
Protecting your telomeres through lifestyle interventions offers a range of evidence-based benefits that extend far beyond just slowing one biomarker. These strategies improve overall cellular health, reduce disease risk, and support healthy aging across multiple body systems.
Can Exercise Really Slow Telomere Shortening?
Regular aerobic exercise is arguably the single most powerful telomere protector available. Studies consistently show that physically active individuals have longer telomeres—equivalent to being approximately 9 years younger biologically—compared to sedentary peers. Exercise increases telomerase activity, reduces oxidative stress, improves mitochondrial function, and lowers systemic inflammation ([10]).
The sweet spot appears to be 30–40 minutes of moderate aerobic exercise, 5 times per week, combined with 2–3 sessions of resistance training. Even modest amounts of walking provide measurable benefits.
Does Stress Management Actually Affect Your Telomeres?
Yes—and the evidence is striking. A landmark study by Epel and Blackburn found that mothers caring for chronically ill children had telomeres equivalent to 10 additional years of aging compared to controls ([5]). Chronic stress elevates cortisol, which increases oxidative damage and reduces telomerase activity.
Mindfulness-Based Stress Reduction (MBSR) programs have been shown to increase telomerase activity after just 8 weeks, and regular meditation practice is associated with longer telomere maintenance over time.
How Does the Mediterranean Diet Support Telomere Health?
Adherence to a Mediterranean-style diet—rich in vegetables, fruits, whole grains, fish, olive oil, and nuts—is consistently associated with longer telomeres. A large study from the Nurses' Health Study found that greater Mediterranean diet adherence correlated with longer leukocyte telomere length ([9]). The mechanisms involve the diet's anti-inflammatory, antioxidant-rich, and omega-3-heavy profile.
Can Omega-3 Fatty Acids Protect Telomeres?
Multiple studies link higher omega-3 (EPA and DHA) levels to longer telomeres and slower telomere attrition. A 2010 study found that patients with the highest omega-3 blood levels experienced the slowest rate of telomere shortening over five years ([8]). Supplementing with 1,000–2,000 mg of EPA+DHA daily—or eating fatty fish 2–3 times per week—is a practical strategy.
Are There Any Risks or Downsides to Telomere-Focused Strategies?
While lifestyle strategies to protect telomeres are overwhelmingly safe, there are important caveats and limitations to understand. Overhyping telomere science or pursuing aggressive telomere interventions without medical guidance carries real risks.
First, the correlation-versus-causation issue remains unresolved. Having short telomeres doesn't guarantee disease, and having long telomeres doesn't guarantee health. Telomere length is just one biomarker among many. Obsessing over a single measurement can lead to unnecessary anxiety and poorly informed health decisions.
Second, aggressive telomerase activation is genuinely dangerous. Because 85–95% of cancers rely on reactivated telomerase for unlimited growth, any pharmaceutical or supplement claiming to dramatically boost telomerase should be viewed with extreme caution. Modest, lifestyle-driven increases are safe. Supplement-driven telomerase activators (like TA-65, marketed as a telomerase booster) lack robust clinical evidence and carry theoretical cancer risk.
Third, commercial telomere testing has significant limitations. Tests cost $100–$500, show high test-to-test and lab-to-lab variability, and most results aren't clinically actionable. Elizabeth Blackburn herself has noted that testing isn't necessary for most people—the lifestyle recommendations remain the same regardless of your telomere length ([1]).
Finally, there is substantial individual variation. Genetics determine 30–80% of your telomere length, meaning some people have naturally shorter or longer telomeres regardless of lifestyle. The goal isn't to achieve a specific telomere length—it's to optimize your telomeres within your genetic potential.
How Do You Build a Practical Telomere-Protection Protocol?
Protecting your telomeres doesn't require expensive tests or exotic supplements. The most effective strategies are proven, accessible, and largely free. Here is a prioritized, evidence-based protocol based on the strongest research available.
- Priority 1: Exercise consistently. Aim for 150–300 minutes of moderate aerobic exercise per week (brisk walking, cycling, swimming) plus 2–3 strength training sessions. Start where you are—even 10 minutes daily provides measurable benefits. Consistency matters more than intensity.
- Priority 2: Manage stress actively. Incorporate 10–20 minutes of daily meditation, mindfulness, or deep breathing. Consider an 8-week MBSR program, yoga 2–3 times weekly, or regular time in nature. If you struggle with chronic stress, anxiety, or depression, seek professional support—these conditions directly accelerate telomere shortening.
- Priority 3: Optimize sleep. Target 7–9 hours of quality sleep per night with a consistent schedule. Both too little sleep (under 6 hours) and poor sleep quality are linked to shorter telomeres. Address any sleep disorders like apnea or insomnia.
- Priority 4: Adopt a Mediterranean-style diet. Emphasize vegetables, fruits, whole grains, fatty fish, olive oil, and nuts. Minimize processed foods, added sugar, and trans fats. Aim for 5–9 servings of colorful produce daily.
- Priority 5: Supplement strategically. Consider omega-3s (1,000–2,000 mg EPA+DHA daily) and vitamin D (1,000–2,000 IU daily, or more if deficient—aim for 40–60 ng/mL). Eat antioxidant-rich foods like berries, green tea, and dark chocolate before reaching for supplement pills.
- Priority 6: Build social connections. Loneliness and social isolation are associated with shorter telomeres. Maintain meaningful relationships, stay involved in community activities, and cultivate a sense of purpose.
- Priority 7: Eliminate accelerators. Quit smoking (the single most damaging habit for telomeres), limit alcohol, maintain a healthy weight, and reduce exposure to environmental toxins where feasible.
The 80/20 approach works well here. You don't need perfection—focus on the highest-impact strategies (exercise, stress management, diet) and build sustainable habits over months and years.
What Diet and Lifestyle Changes Best Support Telomere Health?
The strongest telomere-protective evidence centers on a combination of dietary patterns, specific nutrients, and daily lifestyle habits that work synergistically to reduce inflammation, lower oxidative stress, and support cellular repair.
The Mediterranean diet pattern consistently shows the strongest association with telomere length in population studies. Key components include:
- Fatty fish 2–3 times per week (salmon, sardines, mackerel for omega-3s)
- Abundant colorful vegetables and fruits (antioxidants, polyphenols)
- Extra virgin olive oil as primary fat (anti-inflammatory oleic acid)
- Nuts and seeds daily (vitamin E, selenium, healthy fats)
- Whole grains over refined (fiber, B vitamins)
- Minimal processed foods, sugar, and red meat
Key nutrients for telomere support:
- Omega-3 fatty acids: EPA and DHA from fish or supplements—the most evidence-backed nutrient for telomere protection
- Vitamin D: Deficiency is linked to shorter telomeres; maintain levels at 40–60 ng/mL
- Vitamin C and E: Antioxidants that protect against oxidative telomere damage
- Polyphenols: Found in green tea (EGCG), berries, and dark chocolate
- Folate: Supports DNA methylation and repair
The Dean Ornish Protocol (Landmark Evidence):
The most compelling evidence for lifestyle-driven telomere changes comes from Dean Ornish's research, published in The Lancet Oncology in 2013. Participants followed a comprehensive program including a plant-based diet, 30 minutes of walking 6 days per week, 60 minutes daily of stress management (yoga, meditation, breathing), and weekly social support groups. After 5 years, the intervention group showed increased telomere length while the control group's telomeres shortened—the first controlled trial demonstrating that lifestyle changes can lengthen telomeres ([7]).
The key lesson from this study? Comprehensive changes work better than single interventions. Greater adherence to the program correlated with greater telomere lengthening.
What Should You Do First to Start Protecting Your Telomeres?
You don't need to overhaul your entire life overnight. The most effective approach is to start with one or two high-impact changes, build consistency, and add more over time. Here's a phased action plan based on the strongest evidence.
Phase 1 (Weeks 1–4): Foundation
- [ ] Start a daily walking habit—aim for 20–30 minutes of brisk walking, 5 days per week
- [ ] Begin a 10-minute daily meditation or deep breathing practice (use a free app like Insight Timer)
- [ ] Add one serving of fatty fish per week (salmon, sardines, or mackerel)
- [ ] Replace one processed snack daily with a handful of nuts or berries
Phase 2 (Weeks 5–8): Build
- [ ] Increase exercise to 40–45 minutes, adding variety (cycling, swimming, or strength training)
- [ ] Expand to a Mediterranean-style eating pattern—more vegetables, olive oil, whole grains
- [ ] Start an omega-3 supplement if not eating fish regularly (1,000 mg EPA+DHA daily)
- [ ] Prioritize sleep: establish a consistent bedtime, aim for 7–9 hours
Phase 3 (Months 3–6): Optimize
- [ ] Check and optimize vitamin D levels (ask your doctor for a blood test)
- [ ] Add strength training 2–3 times per week
- [ ] Deepen stress management—consider an MBSR course, yoga, or therapy if needed
- [ ] Strengthen social connections—schedule regular time with friends, family, or community groups
- [ ] Eliminate or reduce smoking, excessive alcohol, and processed food
Phase 4 (Ongoing): Maintain
- [ ] Sustain 150–300 minutes of weekly exercise (aerobic + resistance)
- [ ] Continue Mediterranean-style eating with minimal processed food
- [ ] Maintain stress management practices daily
- [ ] Focus on the 80/20 rule—consistent good habits matter more than occasional perfection
Frequently asked questions
What are telomeres in simple terms?
Telomeres are protective caps at the ends of your chromosomes, similar to the plastic tips on shoelaces. They prevent your DNA from unraveling or fusing with other chromosomes during cell division. Each time a cell divides, telomeres get slightly shorter, which is one reason our cells age over time. When telomeres become critically short, cells stop dividing properly or die.
Can you actually reverse telomere shortening?
Research suggests you can slow and potentially reverse some telomere shortening through lifestyle changes. The Dean Ornish study showed that comprehensive lifestyle interventions—plant-based diet, exercise, stress management, and social support—led to measurable telomere lengthening over 5 years compared to controls. However, this doesn't mean you can fully reverse decades of aging, and results vary between individuals.
How much does chronic stress affect telomere length?
Chronic stress has a substantial impact on telomere shortening. A landmark study found that mothers caring for chronically ill children had telomeres equivalent to 10 additional years of aging compared to low-stress controls. Stress increases cortisol, oxidative damage, and inflammation—all of which accelerate telomere erosion. Stress management through meditation or therapy can help counteract this effect.
Is telomere testing worth the money?
For most people, telomere testing is not worth the cost. Commercial tests ($100–$500) have high variability between measurements and limited clinical actionability. Your lifestyle recommendations—exercise, healthy diet, stress management, quality sleep—remain the same regardless of your test results. Elizabeth Blackburn herself has said testing isn't necessary for most people. It may interest biohackers or those tracking changes after major lifestyle shifts.
What is the best exercise for protecting telomeres?
Aerobic exercise shows the strongest evidence for telomere protection. Running, cycling, swimming, and brisk walking for 30–40 minutes, 5 times per week, are associated with significantly longer telomeres. A 2019 study in the European Heart Journal found that endurance and HIIT training increased telomerase activity, while resistance training alone did not show the same telomere benefit—though strength training remains important for overall longevity.
Do omega-3 supplements really help telomeres?
Yes, the evidence is reasonably strong. A study published in JAMA found that patients with the highest omega-3 blood levels had the slowest rate of telomere shortening over 5 years. The anti-inflammatory and antioxidant properties of EPA and DHA appear to protect telomeric DNA from oxidative damage. Aim for 1,000–2,000 mg of combined EPA+DHA daily through fish or supplements.
At what age do telomeres start shortening significantly?
Telomere shortening begins at birth and continues with every cell division throughout life. The rate of shortening is fastest during early childhood and then slows to a more gradual decline. However, accelerated shortening can occur at any age due to chronic stress, smoking, poor diet, or sedentary behavior. The good news is that lifestyle interventions can slow the rate of shortening at any age.
Can diet alone protect your telomeres?
Diet is an important piece of the puzzle, but research suggests a comprehensive approach works best. The Mediterranean diet is associated with longer telomeres, and specific nutrients like omega-3s and antioxidants offer protection. However, the Dean Ornish study showed that combining diet with exercise, stress management, and social support produced the strongest results. Think of diet as one powerful lever among several.
Is telomerase activation safe?
Modest, lifestyle-driven increases in telomerase activity appear safe and beneficial. Exercise, meditation, and healthy diet can boost telomerase by up to 30% without observed cancer risk. However, aggressive pharmacological telomerase activation is risky because 85–95% of cancers depend on reactivated telomerase for unlimited growth. Supplements claiming to dramatically boost telomerase lack robust evidence and may carry theoretical cancer risk.
How long does it take to see telomere benefits from lifestyle changes?
Increased telomerase activity can be measured within weeks to months of starting lifestyle changes. Measurable improvements in telomere length typically require months to 1–2 years of consistent effort. Long-term benefits—reduced disease risk and improved healthspan—accumulate over years to decades. Consistency matters far more than intensity. Start with one or two changes and build from there.