longevity
Senescent Cells & Aging | Zombie Cells Explained
Senescent cells (zombie cells) drive aging and disease. Learn about cellular senescence, SASP, senolytics (fisetin, quercetin), and how to clear them.
Somewhere inside you right now, cells are refusing to die.
They stopped dividing months or years ago. They're not functioning properly. But they won't go through apoptosis—the programmed cell death process that normally clears out damaged cells. Instead, they sit there. Consuming resources. Leaking inflammatory chemicals into surrounding tissue. Turning neighboring healthy cells into more zombie cells.
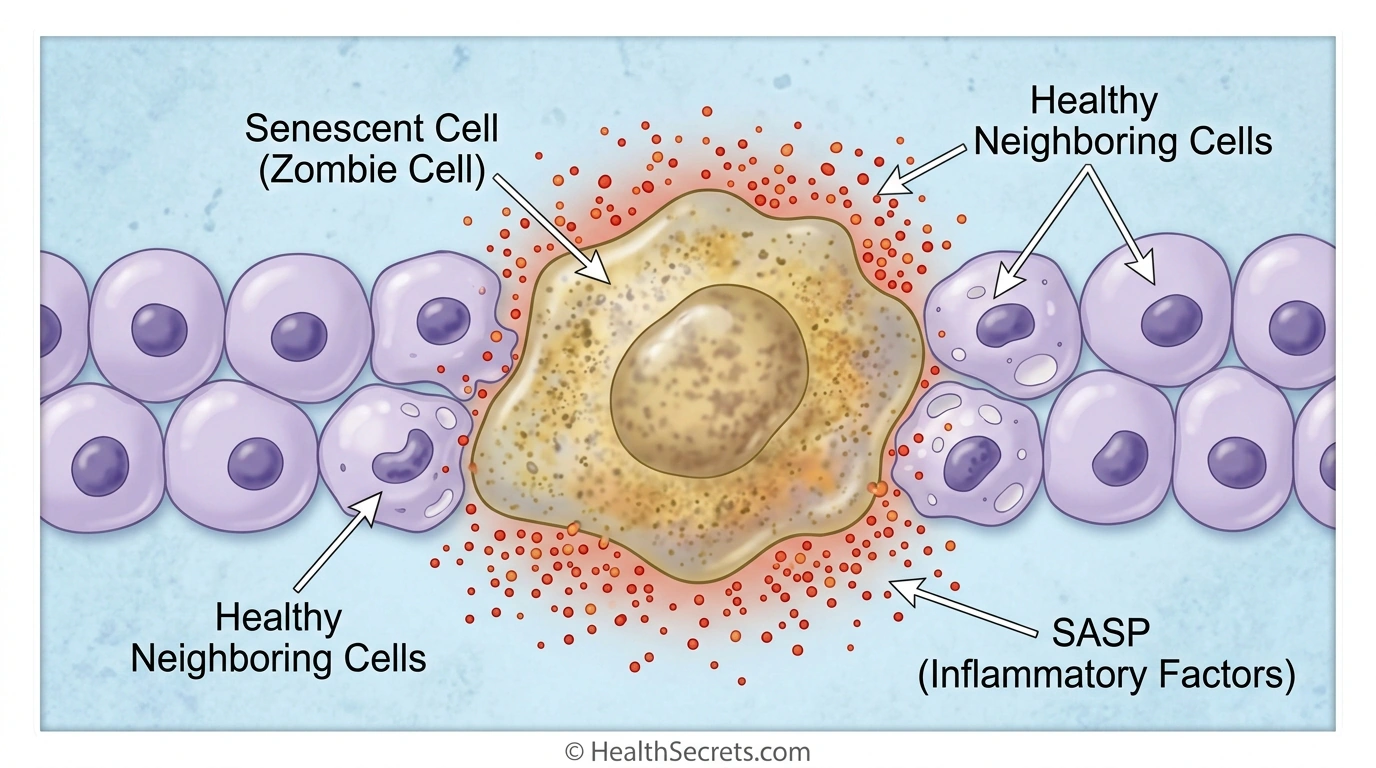
Senescent cells. The scientific community calls them that. Most longevity researchers just call them zombie cells, and honestly, the nickname fits perfectly.
Leonard Hayflick discovered in 1961 that human cells have a built-in division limit—roughly 40 to 60 divisions before they permanently stop. For decades, nobody thought much of it. Cells get old, cells stop dividing, life goes on. But starting in the late 2000s, researchers uncovered something alarming: these cells don't just retire quietly. They actively sabotage the body.
The breakthrough came in 2011 when scientists at the Mayo Clinic proved that clearing senescent cells from mice dramatically improved their health and extended their lifespan. That single study launched an entirely new field of aging research.
In this guide, you'll learn exactly what senescent cells are, how they drive aging through the SASP (senescence-associated secretory phenotype), the landmark studies that changed everything, and what you can realistically do about them today—from lifestyle changes to emerging senolytic supplements like quercetin and fisetin. We'll also cover where the science actually stands versus the hype, because there's plenty of both. If you're exploring the broader landscape of aging interventions, our longevity and anti-aging pillar guide provides additional context, and understanding inflammation's role in chronic disease connects directly to the SASP mechanisms we'll discuss here.
- Senescent cells are damaged cells that stop dividing but refuse to die, accumulating in tissues as you age and reaching 10–15% of cells in older adults
- The SASP (senescence-associated secretory phenotype) is how zombie cells poison neighbors—secreting inflammatory cytokines, proteases, and growth factors that damage surrounding tissue
- Landmark studies by Baker et al. (2011, 2016) proved that clearing senescent cells in mice extended healthspan by 20–30% and delayed multiple age-related diseases
- Senolytics are drugs that selectively kill senescent cells—dasatinib plus quercetin (D+Q) is the most studied combination in human clinical trials
- Fisetin and quercetin are available as over-the-counter supplements with emerging senolytic properties, though optimal dosing protocols remain experimental
- Pulsed dosing (3 days on, 4–8 weeks off) appears more effective and safer than daily supplementation for senolytic compounds
- Exercise, intermittent fasting, and anti-inflammatory diets reduce senescent cell accumulation through natural immune clearance and autophagy
- Human clinical trials are ongoing for conditions including idiopathic pulmonary fibrosis, Alzheimer's disease, and diabetic kidney disease—early results are promising but limited
- Senolytic therapy is experimental, not a replacement for proven lifestyle interventions—optimize exercise, diet, sleep, and stress management first
What Are Senescent Cells and Why Do Scientists Call Them Zombie Cells?
Senescent cells are damaged or aged cells that have permanently stopped dividing but resist programmed cell death, remaining metabolically active while secreting harmful inflammatory compounds into surrounding tissue. They accumulate with age as immune clearance declines, reaching up to 10–15% of cells in older tissues and driving chronic inflammation linked to age-related diseases.
The zombie cell analogy isn't just catchy marketing. It's genuinely accurate.
Think about what zombies do in movies. They're not alive—not functioning, not contributing, not doing anything useful. But they're not dead either. They wander around causing damage and, critically, they convert healthy people into more zombies.
Senescent cells do exactly this. They've stopped performing their normal cellular functions. They're consuming glucose and oxygen without contributing to tissue health. And through the SASP—which we'll get into shortly—they actively convert neighboring healthy cells into more senescent cells.
Leonard Hayflick's 1961 discovery established that normal human cells divide approximately 40–60 times before entering permanent growth arrest ([1]). This "Hayflick limit" was initially considered simply the endpoint of cellular lifespan. Nothing particularly alarming about it.
But the picture changed dramatically when researchers discovered that senescent cells don't just sit there passively. They're metabolically hyperactive. They secrete hundreds of different proteins, lipids, and other molecules that collectively wreak havoc on surrounding tissue ([4]).
In a young, healthy body, this isn't a crisis. Your immune system—particularly natural killer cells and macrophages—identifies and clears senescent cells efficiently. A few zombie cells appear, the immune system takes them out, balance is maintained.
The problem emerges with aging. Immune surveillance declines. Senescent cells accumulate faster than the immune system can remove them. And each senescent cell creates conditions that generate more senescent cells. The balance tips, and the zombie invasion begins in earnest.
How Does Cellular Senescence Actually Develop and Spread Through Your Body?
Cellular senescence develops when cells experience severe stress or damage—from telomere shortening, DNA mutations, oxidative stress, or oncogene activation—triggering permanent growth arrest as a protective mechanism against cancer. The process becomes harmful when the SASP creates a feedback loop: senescent cells secrete inflammatory factors that damage neighbors and induce more senescence.
Cells don't randomly decide to become senescent. Specific triggers push them there, and understanding these triggers explains why senescent cell accumulation accelerates with age.
What Triggers Cells to Become Senescent?
- Telomere shortening is the classic trigger. Every cell division shortens the protective caps on your chromosomes slightly. After roughly 50 divisions, telomeres become critically short, and the cell enters senescence permanently. This is the Hayflick limit in action—a built-in replication counter.
- DNA damage from radiation, environmental toxins, or oxidative stress can push cells into senescence at any point. The cell detects severe damage and shuts down division rather than risk passing mutations to daughter cells. It's a cancer prevention mechanism. Better a zombie cell than a tumor.
- Oxidative stress from excessive free radicals overwhelms cellular repair capacity.
- Oncogene activation—when cancer-promoting genes switch on inappropriately—triggers senescence as an emergency brake.
- Mitochondrial dysfunction creates a vicious cycle where damaged mitochondria produce more reactive oxygen species, generating more damage.
And chronic inflammation itself drives senescence—which matters enormously, because senescent cells then produce more inflammation.
How Does the SASP Turn One Zombie Cell Into Many?
The SASP is where senescent cells go from annoying to genuinely destructive.
Senescent cells secrete a toxic cocktail: pro-inflammatory cytokines like IL-6 and IL-8, chemokines that attract immune cells, matrix metalloproteinases that break down tissue structure, growth factors that disrupt normal cellular behavior, and reactive oxygen species that cause oxidative damage ([5]).
Originally, this secretory program serves a purpose. It's the senescent cell shouting to the immune system: come clear me out. In young organisms, immune cells respond, remove the senescent cell, and tissue homeostasis is restored.
But in older organisms with declining immune function? Nobody answers the call. The senescent cell keeps secreting inflammatory factors. Those factors damage neighboring cells. Damaged neighbors become senescent themselves. Those new senescent cells begin secreting their own SASP factors.
A 2024 review in Nature Reviews Molecular Cell Biology detailed how the SASP drives what researchers call "inflammaging"—the chronic, low-grade inflammation that characterizes biological aging and underlies virtually every age-related disease ([10]).
The feedback loop accelerates: more senescent cells → more SASP → more inflammation → more cellular damage → more senescent cells. Breaking this cycle is the entire premise of senolytic therapy.
What Are the Proven Benefits of Clearing Senescent Cells?
Clearing senescent cells from aging mice extended healthspan by 20–30%, delayed the onset of cancer, preserved kidney and heart function, maintained muscle mass, and improved metabolic health in landmark studies. These results demonstrated that senescent cell accumulation is not just a consequence of aging but an active driver that can be therapeutically targeted.
The field transformed because of a handful of studies that nobody could ignore.
What Did the Baker 2011 Study Prove About Senescent Cells and Aging?
In 2011, Darren Baker and Jan van Deursen at the Mayo Clinic genetically engineered mice to allow selective elimination of cells expressing p16Ink4a—a key senescence marker. When they cleared these cells from middle-aged mice, the results were striking. Delayed cataracts. Preserved muscle mass. Reduced fat tissue dysfunction. Improved overall healthspan ([2]).
This was the proof of concept. Senescent cells aren't passive bystanders in aging—they're active participants, and removing them improves outcomes.
The 2016 follow-up was even more dramatic. Lifelong clearance of senescent cells extended median lifespan by roughly 25%, prevented multiple age-related pathologies simultaneously, and the treated mice were healthier across virtually every measure ([3]).
In 2018, Bussian and colleagues showed that clearing senescent glial cells from Alzheimer's mouse models reduced tau pathology and neuroinflammation—suggesting senescent cells contribute directly to neurodegeneration ([8]).
These aren't marginal improvements. Clearing senescent cells in animals consistently produces dramatic, multi-system health benefits. The question that remains—and it's a big one—is whether the same approach works in humans.
What Are the Risks and Unknowns of Targeting Senescent Cells?
The primary risks include unknown long-term safety effects, potential disruption of beneficial senescence functions like tumor suppression and wound healing, uncertain optimal dosing protocols, and limited human clinical data. Senescent cells serve protective roles in cancer prevention and tissue repair, so eliminating them indiscriminately could theoretically cause harm.
I want to be honest about this: the excitement around senolytics sometimes outpaces the evidence.
Senescence isn't entirely bad. It's originally a cancer prevention mechanism. When a cell accumulates dangerous mutations, entering senescence stops it from becoming a tumor. Acute senescence during wound healing helps coordinate tissue repair. During embryonic development, senescence plays essential roles in organ formation.
The problem is chronic senescence—cells that linger for months or years, pumping out SASP factors. The distinction between beneficial acute senescence and harmful chronic senescence matters, and current senolytic approaches don't always differentiate between them.
Other specific concerns:
- Long-term safety data doesn't exist. The oldest human senolytic trials are only a few years old. We don't know what decades of periodic senescent cell clearance does to the body
- Bioavailability is uncertain. Oral quercetin and fisetin supplements may not reach therapeutic concentrations in all tissues
- Drug interactions. Quercetin has mild anticoagulant properties and can interact with certain medications
- Individual variation. What works in clinical trial averages may not apply to every individual
- The 2026 NIA bone health trial found that senolytic therapy had only subtle impact on bone biomarkers—a reminder that dramatic mouse results don't always translate directly to humans ([14])
None of this means senolytics are dangerous. Quercetin and fisetin have generally good safety profiles from decades of use as supplements. But treating them as proven anti-aging interventions oversells where the science currently stands.
How Can You Actually Address Senescent Cell Accumulation Today?
The most evidence-backed approach combines proven lifestyle interventions—regular exercise, intermittent fasting, and anti-inflammatory nutrition—with cautious experimental use of senolytic supplements like quercetin or fisetin in pulsed dosing protocols (3 days on, 4–8 weeks off) for adults over 60 or those with age-related conditions, always under medical guidance.
What Are Senolytics and How Do They Work?
Senolytics target the anti-apoptotic pathways that senescent cells use to resist programmed cell death. Normal cells don't rely as heavily on these survival pathways, so senolytics selectively push senescent cells over the edge while leaving healthy cells intact.
Dasatinib plus quercetin (D+Q) remains the most studied combination. Dasatinib is a prescription cancer drug (tyrosine kinase inhibitor); quercetin is a plant flavonoid. Together, they target different anti-apoptotic pathways and are more effective than either alone ([6]).
Human trials of D+Q are testing outcomes in idiopathic pulmonary fibrosis ([11]), diabetic kidney disease, Alzheimer's disease ([12]), and frailty. A 2024 study found D+Q influenced epigenetic aging clocks—suggesting senolytics may affect biological aging at the molecular level ([13]).
Fisetin—a flavonoid concentrated in strawberries—emerged as a potent senolytic in Yousefzadeh's landmark 2018 study, extending health and lifespan in mice even when treatment started in old age ([7]).
Should You Consider Senolytic Supplements?
If you're over 60, dealing with age-related conditions, and have already optimized your lifestyle foundations—maybe. The risk-benefit calculation shifts with age and health status.
If you're a healthy 30-year-old? Probably not worth it yet. Your immune system is still clearing senescent cells effectively, and the potential benefits don't clearly outweigh the unknowns.
If you decide to experiment:
- Use pulsed dosing: 1,000–2,000mg quercetin or fisetin for 3 consecutive days, then 4–8 weeks off
- Take with dietary fat (improves absorption significantly)
- Monitor for GI side effects (most common complaint)
- Combine with lifestyle optimization—supplements aren't a substitute
- Discuss with your healthcare provider, especially if you take medications
Which Foods and Lifestyle Habits Help Reduce Senescent Cell Buildup?
Regular aerobic exercise is the single most effective natural intervention for reducing senescent cell accumulation, supported by intermittent fasting to activate autophagy, an anti-inflammatory Mediterranean-style diet rich in quercetin and fisetin from whole foods (onions, apples, berries, strawberries), and avoidance of accelerators like smoking, chronic stress, and sedentary behavior.
Before spending money on supplements, get the fundamentals right. These lifestyle approaches reduce senescent cell burden through mechanisms your body already has built in.
- Exercise is non-negotiable. Regular aerobic activity reduces senescent cell markers across multiple tissues—likely through improved immune surveillance, reduced systemic inflammation, and enhanced autophagy. You don't need extreme protocols. Consistent moderate activity—150+ minutes per week of brisk walking, cycling, swimming—delivers meaningful benefits.
- Fasting and caloric restriction activate autophagy, your body's cellular cleanup system. Autophagy degrades and recycles damaged cellular components, potentially including senescent cells. Intermittent fasting protocols (16:8 or periodic 24-hour fasts) provide a practical way to activate these pathways.
- Quercetin-rich foods include onions (especially red onions), apples, berries, capers, leafy greens, and tea.
- Fisetin-rich foods include strawberries, apples, persimmons, onions, and cucumbers. You won't reach senolytic doses from food alone, but consistent dietary intake supports anti-inflammatory and antioxidant defenses.
- Green tea's EGCG (epigallocatechin gallate) shows some senolytic activity in research. Three to five cups daily, or 400–800mg supplemental EGCG, may contribute.
- Curcumin from turmeric has weaker senolytic evidence but strong anti-inflammatory properties that reduce the conditions driving cellular senescence.
- What to avoid: Smoking accelerates senescence massively through oxidative stress. Chronic psychological stress promotes inflammation-driven senescence. Excessive alcohol damages cells directly. Poor sleep impairs repair mechanisms. A sedentary lifestyle reduces immune clearance capacity.
The lifestyle foundation matters more than any supplement. Get this right first.
What Is a Practical Action Plan for Addressing Senescent Cells at Any Age?
A phased approach works best: optimize lifestyle foundations first (exercise, nutrition, sleep, stress management), then consider adding evidence-based supplements in pulsed protocols if you're over 50 or managing age-related conditions, and stay informed about emerging clinical trial results that may change recommendations.
Phase 1 — Foundation (Start Now, Any Age):
- [ ] Establish 150+ minutes weekly aerobic exercise (brisk walking, cycling, swimming)
- [ ] Adopt anti-inflammatory eating pattern (Mediterranean-style, rich in colorful fruits and vegetables)
- [ ] Include quercetin-rich foods daily (onions, apples, berries, green tea)
- [ ] Include fisetin-rich foods regularly (strawberries, persimmons)
- [ ] Implement intermittent fasting protocol (16:8 or periodic 24-hour fasts)
- [ ] Optimize sleep quality (7–9 hours, consistent schedule)
- [ ] Eliminate smoking and minimize alcohol intake
Phase 2 — Targeted Supplementation (Age 50+ or Age-Related Conditions):
- [ ] Discuss senolytic supplements with healthcare provider
- [ ] Consider quercetin (1,000mg pulsed: 3 days on, 4–8 weeks off)
- [ ] Consider fisetin (100–500mg pulsed protocol)
- [ ] Add green tea extract EGCG (400–800mg daily) for additional support
- [ ] Take senolytic supplements with dietary fat for improved absorption
- [ ] Monitor for side effects and adjust protocol accordingly
Phase 3 — Stay Informed (Ongoing):
- [ ] Follow clinical trial results for D+Q, fisetin, and next-generation senolytics
- [ ] Discuss emerging therapies with your physician as evidence matures
- [ ] Reassess your protocol annually based on new research
References
- Hayflick, L. and Moorhead, P.S., 1961. The serial cultivation of human diploid cell strains. Experimental Cell Research, 25(3), pp.585–621. Source
- Baker, D.J. et al., 2011. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature, 479(7372), pp.232–236. Source
- Baker, D.J. et al., 2016. Naturally occurring p16Ink4a-positive cells shorten healthy lifespan. Nature, 530(7589), pp.184–189. Source
- van Deursen, J.M., 2014. The role of senescent cells in ageing. Nature, 509(7501), pp.439–446. Source
- Coppé, J.P. et al., 2010. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annual Review of Pathology, 5, pp.99–118. Source
- Zhu, Y. et al., 2015. The Achilles' heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell, 14(4), pp.644–658. Source
- Yousefzadeh, M.J. et al., 2018. Fisetin is a senotherapeutic that extends health and lifespan. EBioMedicine, 36, pp.18–28. Source
- Bussian, T.J. et al., 2018. Clearance of senescent glial cells prevents tau-dependent pathology and cognitive decline. Nature, 562(7728), pp.578–582. Source
- Childs, B.G., Li, H. and van Deursen, J.M., 2018. Senescent cells: a therapeutic target for cardiovascular disease. Journal of Clinical Investigation, 128(4), pp.1217–1228. Source
- SASP Review, 2024. Senescence-associated secretory phenotype in physiological and pathological contexts. Nature Reviews Molecular Cell Biology. Source
- Justice, J.N. et al., 2019. Senolytics in idiopathic pulmonary fibrosis: results from a first-in-human, open-label, pilot study. EBioMedicine, 40, pp.554–563. Source
- Millar, C. et al., 2025. Dasatinib plus quercetin for Alzheimer's disease: a pilot study. Lancet eBioMedicine. Source
Frequently asked questions
What are senescent cells in simple terms?
Senescent cells are damaged cells that have permanently stopped dividing but refuse to die. They're often called zombie cells because—like zombies—they're not functioning normally but remain active, releasing harmful inflammatory chemicals (the SASP) that damage surrounding healthy tissue and accelerate aging. They accumulate in your body as your immune system's ability to clear them declines with age.
Why are senescent cells called zombie cells?
The zombie analogy fits because senescent cells share key zombie characteristics: they're not truly alive (not performing normal cell functions or dividing), they're not dead (still metabolically active and consuming resources), they cause harm to those around them (through SASP inflammatory secretions), and they can convert healthy neighbors into more zombie cells (paracrine senescence). The analogy was popularized by aging researchers to make the concept accessible.
How many senescent cells does a person have?
In young adults, senescent cells represent a tiny fraction of total cells, efficiently cleared by the immune system. By older age (70+), senescent cells can comprise 10–15% of cells in certain tissues. The accumulation varies by tissue type—skin, fat, joints, and lungs tend to accumulate more senescent cells than other organs.
What is the SASP and why does it matter?
The SASP (senescence-associated secretory phenotype) is the collection of inflammatory cytokines, chemokines, proteases, and growth factors that senescent cells release into surrounding tissue. It matters because the SASP drives chronic inflammation (inflammaging), breaks down tissue structure, impairs stem cell function, promotes age-related diseases, and induces senescence in neighboring healthy cells—creating a destructive feedback loop.
What are senolytics and how do they work?
Senolytics are drugs or compounds that selectively kill senescent cells by targeting the anti-apoptotic (anti-death) pathways these cells use to resist programmed cell death. Unlike normal cells, senescent cells are heavily dependent on survival pathways like BCL-2 family proteins. Senolytics disable these survival mechanisms, causing senescent cells to undergo apoptosis while leaving healthy cells unaffected.
Is fisetin a senolytic?
Yes, fisetin has demonstrated senolytic properties in research. Yousefzadeh et al. (2018) showed that fisetin reduced senescent cell burden and extended healthspan and lifespan in mice, even when treatment started in old age. Fisetin appears to work by targeting multiple anti-apoptotic pathways in senescent cells. It's currently available as a dietary supplement and is being studied in human clinical trials for frailty and cognitive decline.
Can you clear senescent cells naturally without supplements?
Yes, several lifestyle interventions help reduce senescent cell accumulation. Regular aerobic exercise improves immune clearance of senescent cells. Intermittent fasting activates autophagy, which helps clear damaged cellular components. An anti-inflammatory diet reduces the oxidative stress that drives cells into senescence. These approaches won't eliminate all senescent cells, but they meaningfully slow accumulation and should be your foundation before considering supplements.
How often should you take senolytic supplements?
Research protocols use pulsed dosing rather than daily supplementation—typically 3 consecutive days of high-dose quercetin (1,000–2,000mg) or fisetin (1,000–1,500mg), followed by 4–8 weeks off. This "hit-and-run" approach works because you don't need continuous senolytic activity; clear the senescent cells, let the body recover, repeat periodically. Daily dosing isn't recommended and may reduce effectiveness.
What is the difference between quercetin and fisetin for senolytics?
Both are plant flavonoids with senolytic properties, but they differ in potency and mechanisms. Fisetin appears to be a more potent senolytic in animal studies, while quercetin is better studied as part of the dasatinib+quercetin (D+Q) combination. Quercetin is more widely available and cheaper. Fisetin has poorer bioavailability but may be more effective per milligram when absorption is optimized through liposomal delivery.
Are senolytic supplements safe?
Quercetin and fisetin have generally good safety profiles based on decades of supplement use. Common side effects include mild GI discomfort. However, long-term safety of senolytic-dose pulsed protocols is unknown because these specific regimens are relatively new. People on blood thinners should use caution with quercetin (mild anticoagulant effects). Pregnant or breastfeeding women should avoid senolytic protocols. Always consult a healthcare provider before starting.